UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER PURSUANT TO RULE 13a-16 OR 15d-16 UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of January, 2026.
Commission File Number: 001-40530
GH Research PLC
(Exact name of registrant as specified in its charter)
Joshua Dawson House
Dawson Street
Dublin 2
D02 RY95
Ireland
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F:
|
Form 20-F
|
☒ |
Form 40-F
|
INFORMATION CONTAINED IN THIS REPORT ON FORM 6-K
On January 5, 2026, GH Research PLC (the “Company”) announced that the U.S. Food and Drug Administration (FDA) has lifted the
clinical hold on GH001, provided further development updates, and made available an updated corporate presentation on its website. A copy of the press release is exhibited hereto as Exhibit 99.1 and a copy of the corporate presentation is
attached hereto as Exhibit 99.2.
The fact that this press release and corporate presentation are being made available should not be deemed an admission as to the
materiality of any information contained in the materials. The information contained in the press release and corporate presentation is being provided as of January 5, 2026, and the Company does not undertake any obligation to update the press
release or the presentation in the future or to update forward-looking statements to reflect subsequent actual results.
1
EXHIBIT INDEX
|
Exhibit No.
|
Description
|
|
Press release dated January 5, 2026
|
|
| 99.2 |
Corporate presentation for January 2026
|
2
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
GH Research PLC
|
||
|
Date: January 5, 2026
|
||
|
By:
|
/s/ Julie Ryan
|
|
|
Name:
|
Julie Ryan
|
|
|
Title:
|
Vice President, Finance
|
|
3
Exhibit 99.1
GH Research Announces FDA Lifts Clinical Hold on GH001, Clearing Path for Global Phase 3 Initiation in 2026
|
•
|
GH001 cleared by FDA for U.S. clinical investigation, enabling U.S. subject enrollment
|
| • |
Company to seek FDA alignment on global Phase 3 program replicating Phase 2b design
|
|
•
|
Phase 3 initiation targeted for 2026
|
Dublin, Ireland, January 5, 2026 – GH Research PLC (Nasdaq: GHRS), a clinical-stage biopharmaceutical company dedicated
to transforming the lives of patients by developing a practice-changing treatment in depression, today announced that the U.S. Food and Drug Administration (FDA) has lifted the clinical hold on its Investigational New Drug Application (IND) for
GH001.
This clearance enables U.S. subject enrollment and progresses the company toward alignment of its development across major jurisdictions.
“The FDA clearance is a major milestone and positions us to advance GH001 as a potential ultra-rapid and durable treatment option for TRD patients,” said Dr.
Velichka Valcheva, Chief Executive Officer. “We continue to expect initiation of our global pivotal program in 2026. We look forward to meeting with the FDA to align on the design for the pivotal Phase 3 program.”
GH001 Phase 2b Profile Highlights from the Previously Reported Phase 2b trial (GH001-TRD-201):
|
•
|
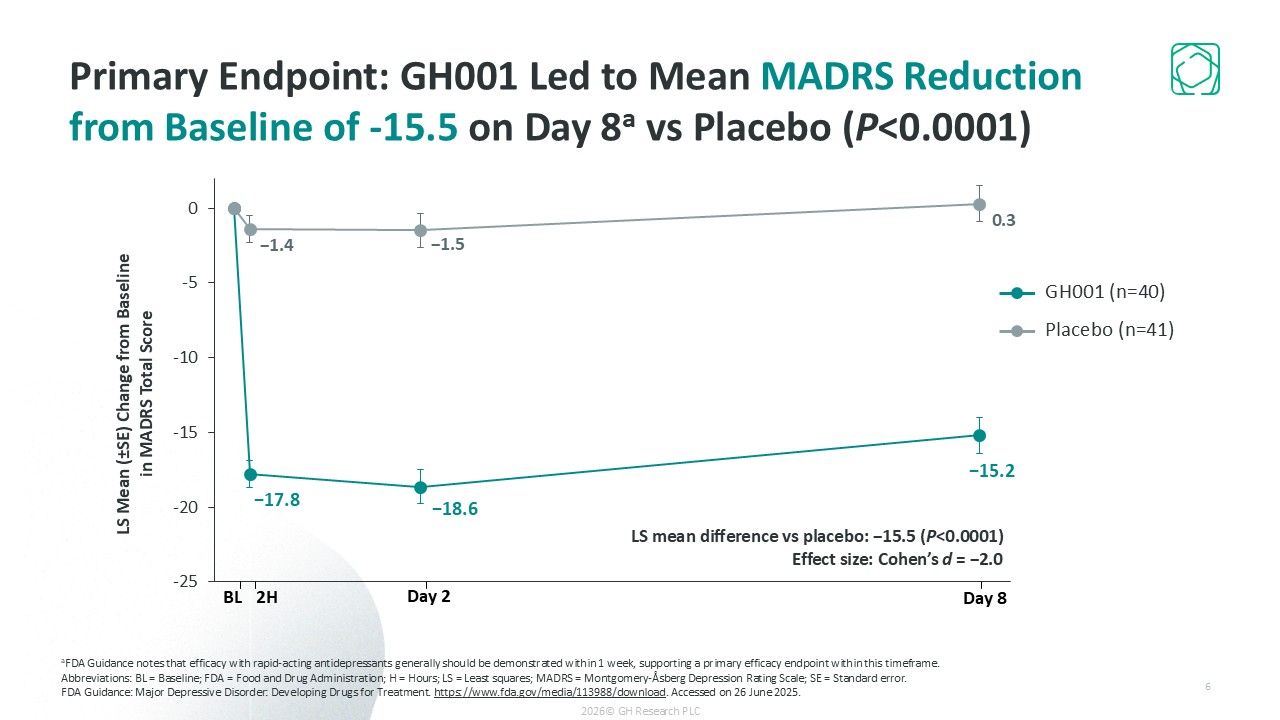
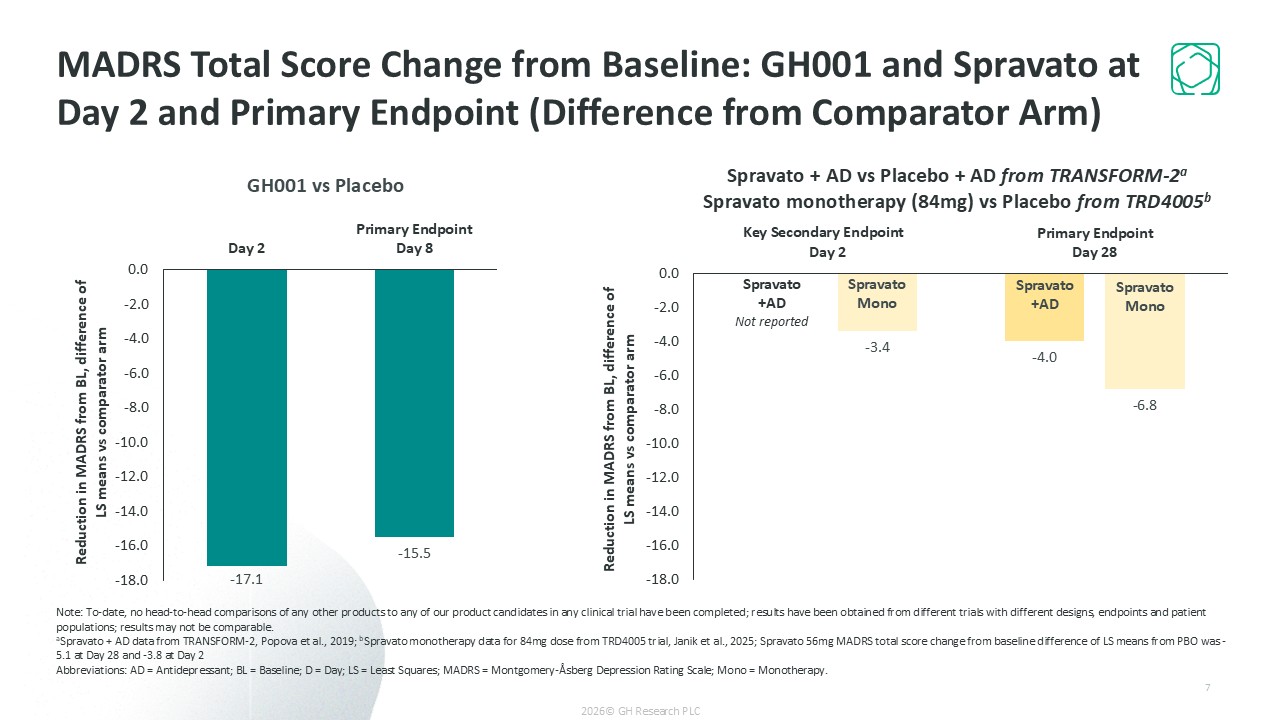
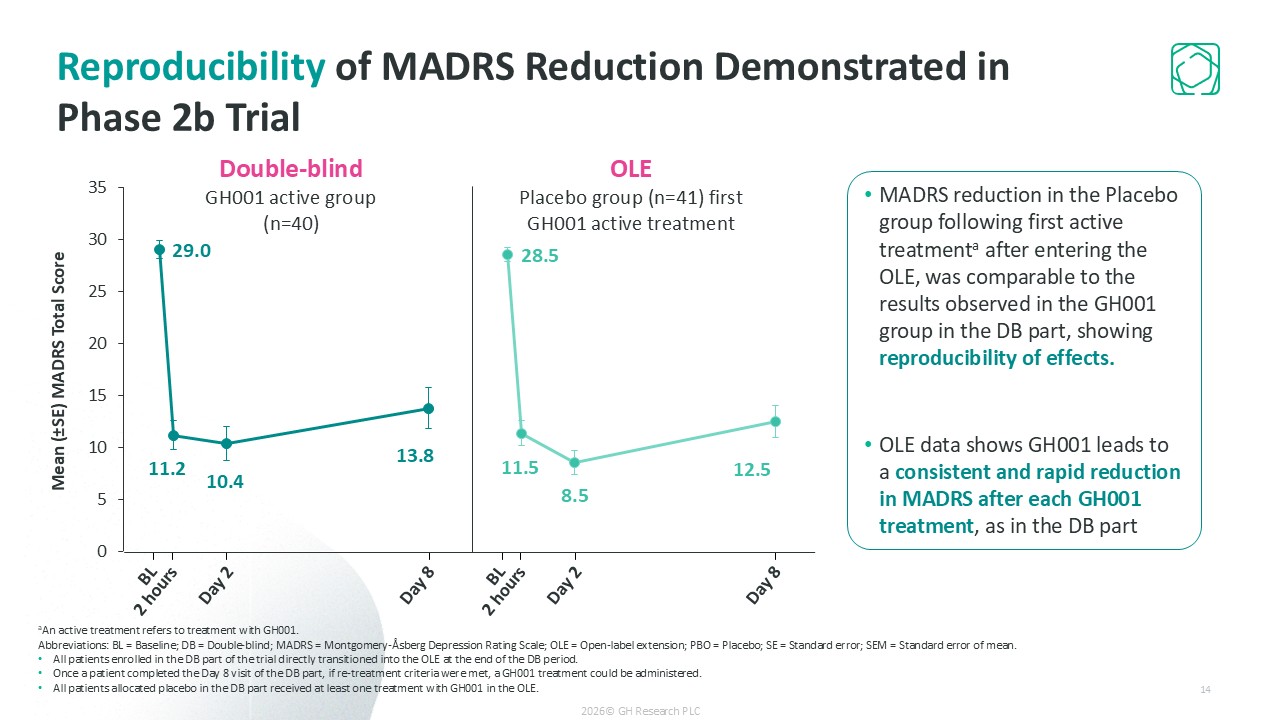
Primary endpoint met: -15.5 point placebo-adjusted MADRS reduction on Day 8 (p<0.0001)
|
|
•
|
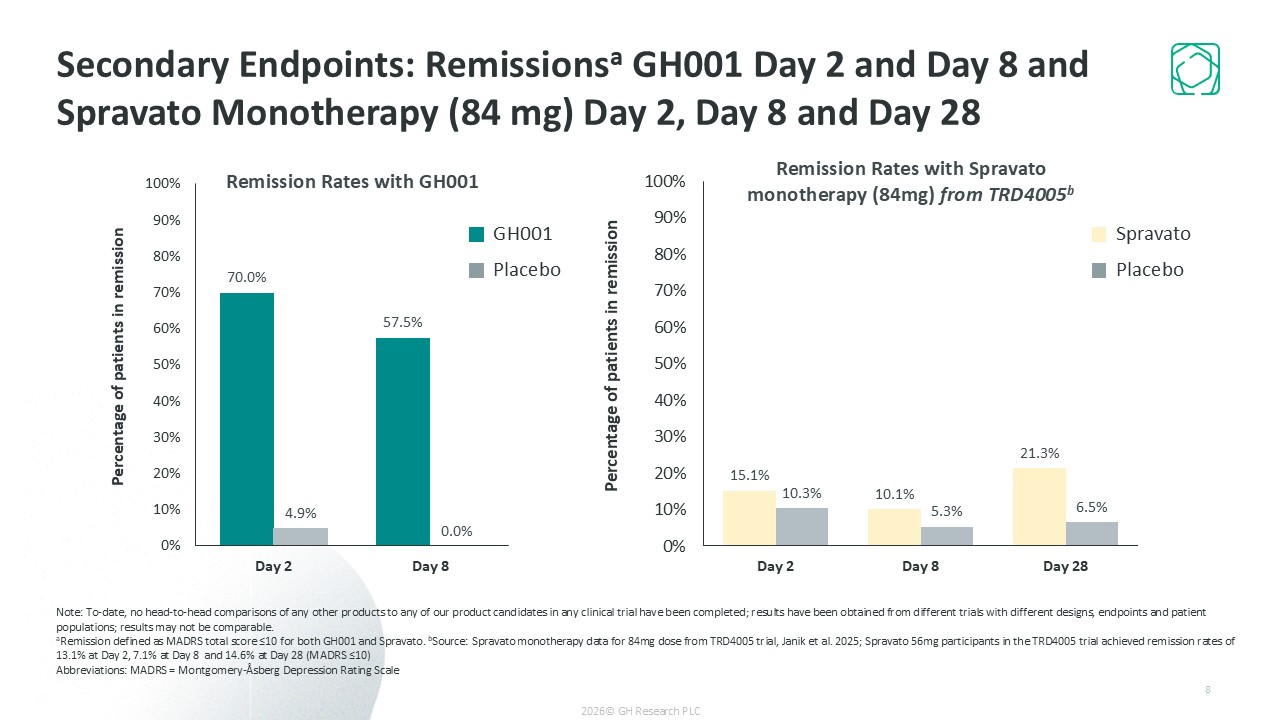
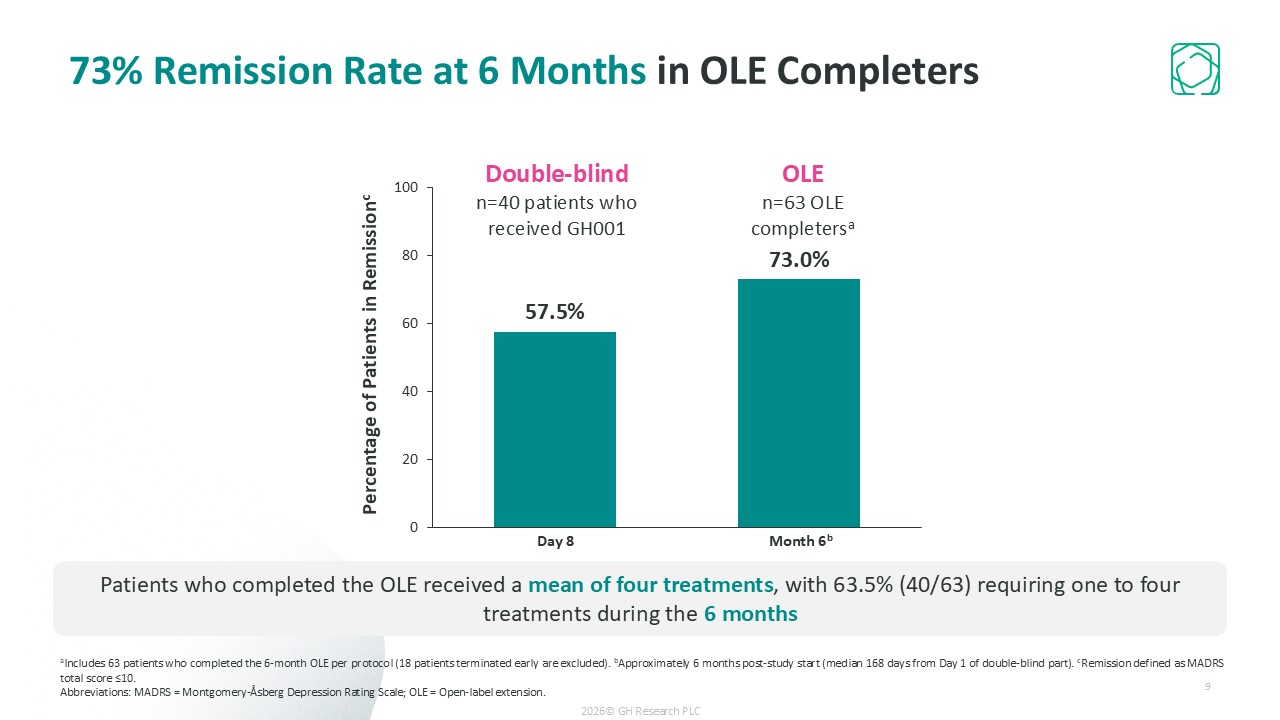
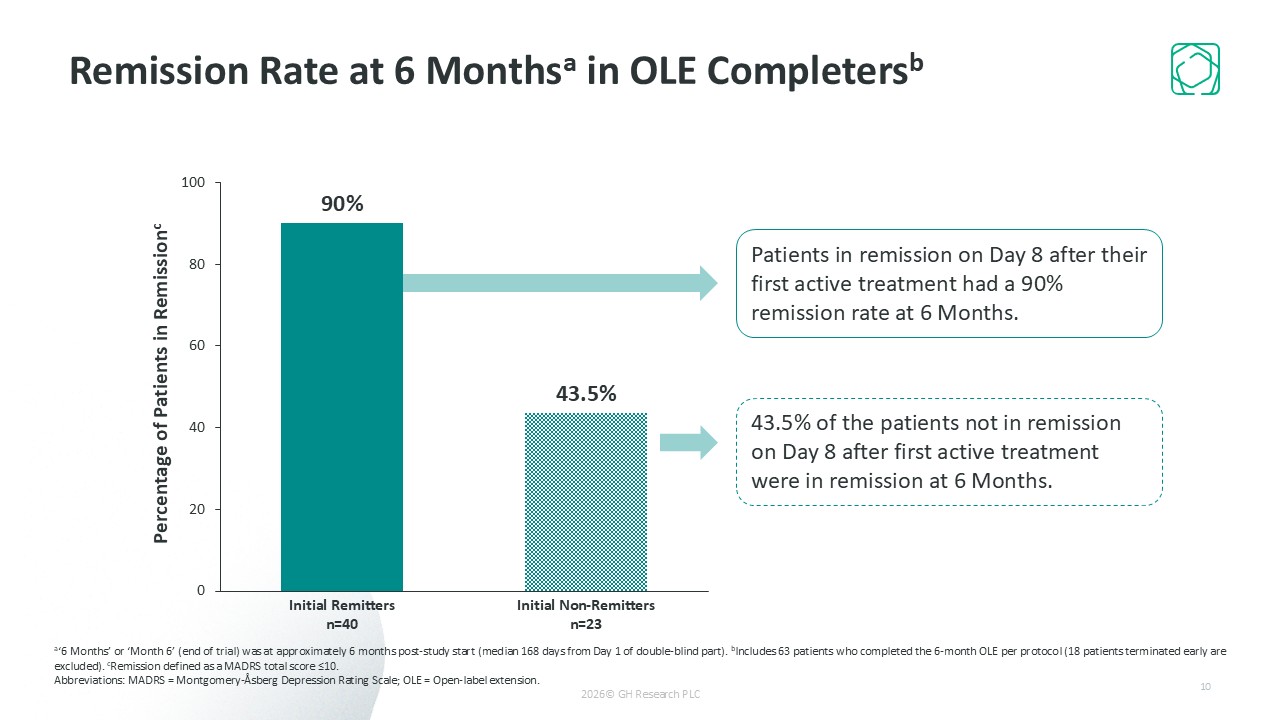
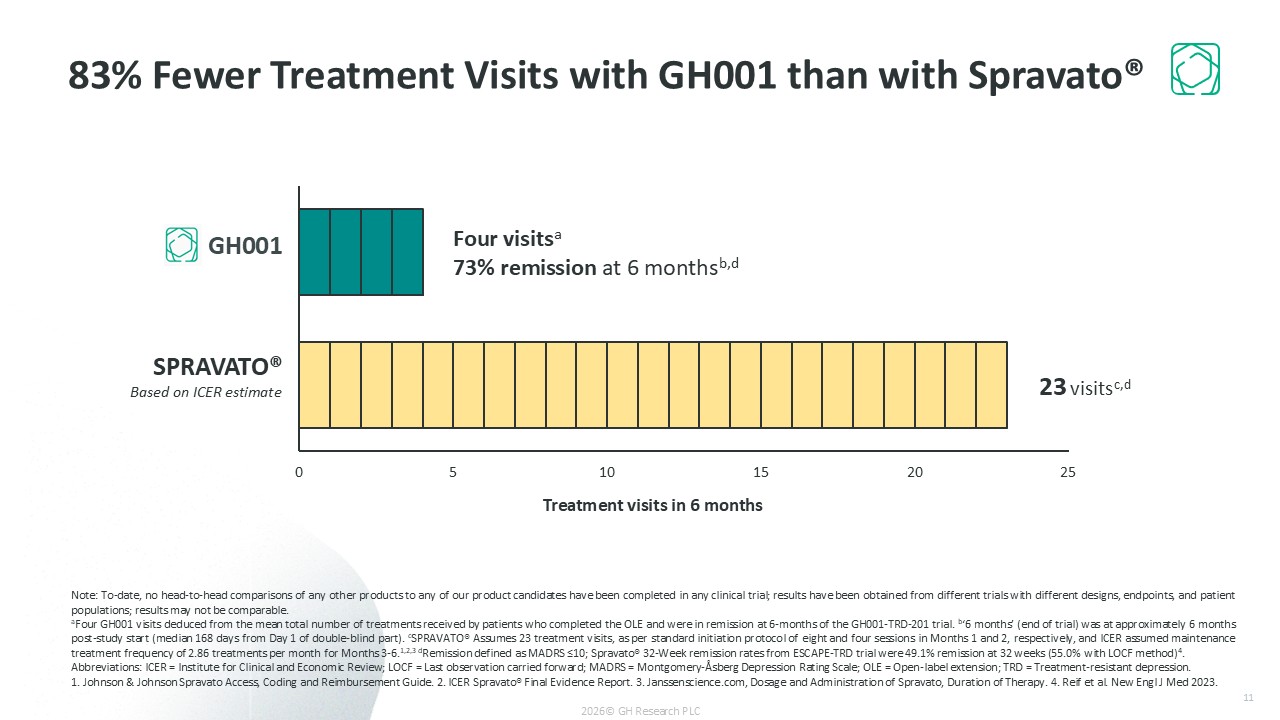
Ultra-rapid remission (57.5% of patients on Day 8); 73% remission at 6 months with infrequent dosing (~4 treatments on average)
|
|
•
|
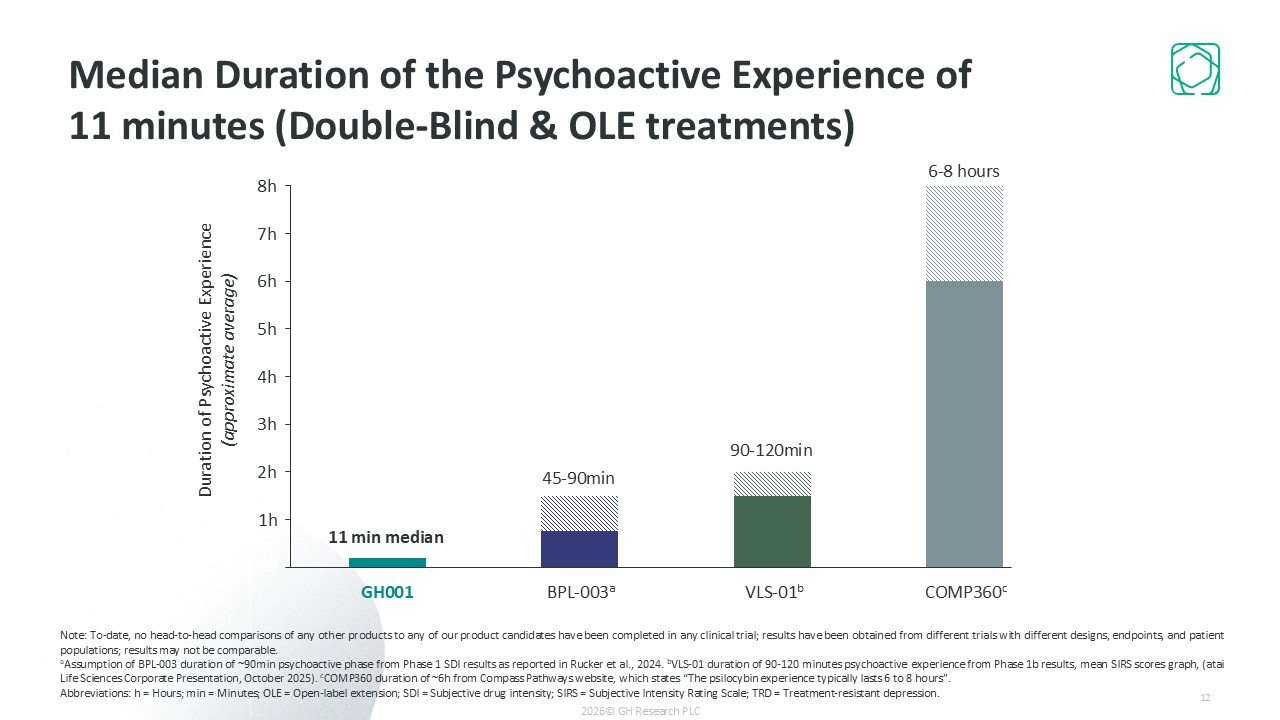
Short psychoactive experience (median of ~11 minutes)
|
| • |
No required psychotherapy
|
|
•
|
99% of patients discharge-ready within 1 hour of dosing
|
|
•
|
Favorable safety: well-tolerated, no serious treatment-related adverse events
|
Recent presentations at scientific conferences reinforce GH001's potential for integration into existing interventional psychiatry practices through its
convenient administration profile.
Michael E. Thase, MD, Professor of Psychiatry, Perelman School of Medicine at the University of Pennsylvania, said, “The large and rapid antidepressant effect
observed with GH001 in the Phase 2b trial, combined with sustained remission through infrequent, short clinic visits, has the potential to be practice-changing for patients with treatment-resistant depression.”
About GH Research PLC
GH Research PLC is a clinical-stage biopharmaceutical company dedicated to transforming the lives of patients by developing a practice-changing treatment in
depression. GH Research PLC's initial focus is on developing its novel and proprietary mebufotenin therapies for the treatment of patients with treatment-resistant depression (TRD).
About GH001
Our lead product candidate, GH001, is formulated for mebufotenin administration via a proprietary inhalation approach. Based on the observed clinical activity
in our Phase 2b GH001-TRD-201 trial, where the primary endpoint was met with a MADRS reduction from baseline of -15.5 points compared with placebo on Day 8 (p<0.0001), we believe that GH001 has the potential to change the way TRD is treated today.
Forward-Looking Statements
This press release contains statements that are, or may be deemed to be, forward-looking statements. All statements other than statements of historical fact
included in this press release, including expectations with regard to initiating our global pivotal program, and the expected timing of such initiation, our future results of operations and financial position, business strategy, product candidates,
research pipeline, ongoing and currently planned preclinical studies and clinical trials, regulatory submissions and approvals, research and development costs, timing and likelihood of success, as well as plans and objectives of management for future
operations, are forward-looking statements. Forward-looking statements appear in a number of places in this press release and include, but are not limited to, statements regarding our intent, belief or current expectations. Forward-looking statements
are based on our management’s beliefs and assumptions and on information currently available to our management. Such statements are subject to risks and uncertainties, and actual results may differ materially from those expressed or implied in the
forward-looking statements due to various factors, including, but not limited to, those described in our filings with the U.S. Securities and Exchange Commission. No assurance can be given that such future results will be achieved. Such
forward-looking statements contained in this document speak only as of the date of this press release. We expressly disclaim any obligation or undertaking to update these forward-looking statements contained in this press release to reflect any
change in our expectations or any change in events, conditions, or circumstances on which such statements are based unless required to do so by applicable law. No representations or warranties (expressed or implied) are made about the accuracy of any
such forward-looking statements.
Investor Relations
Julie Ryan
GH Research PLC
investors@ghres.com
Exhibit 99.2