UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER PURSUANT TO RULE 13a-16 OR 15d-16 UNDER
THE SECURITIES EXCHANGE ACT OF 1934
For the month of January 2026.
Commission File Number: 001-40530
GH Research PLC
(Exact name of registrant as specified in its charter)
Joshua Dawson House
Dawson Street
Dublin 2
D02 RY95
Ireland
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F:
|
Form 20-F
|
☒
|
Form 40-F
|
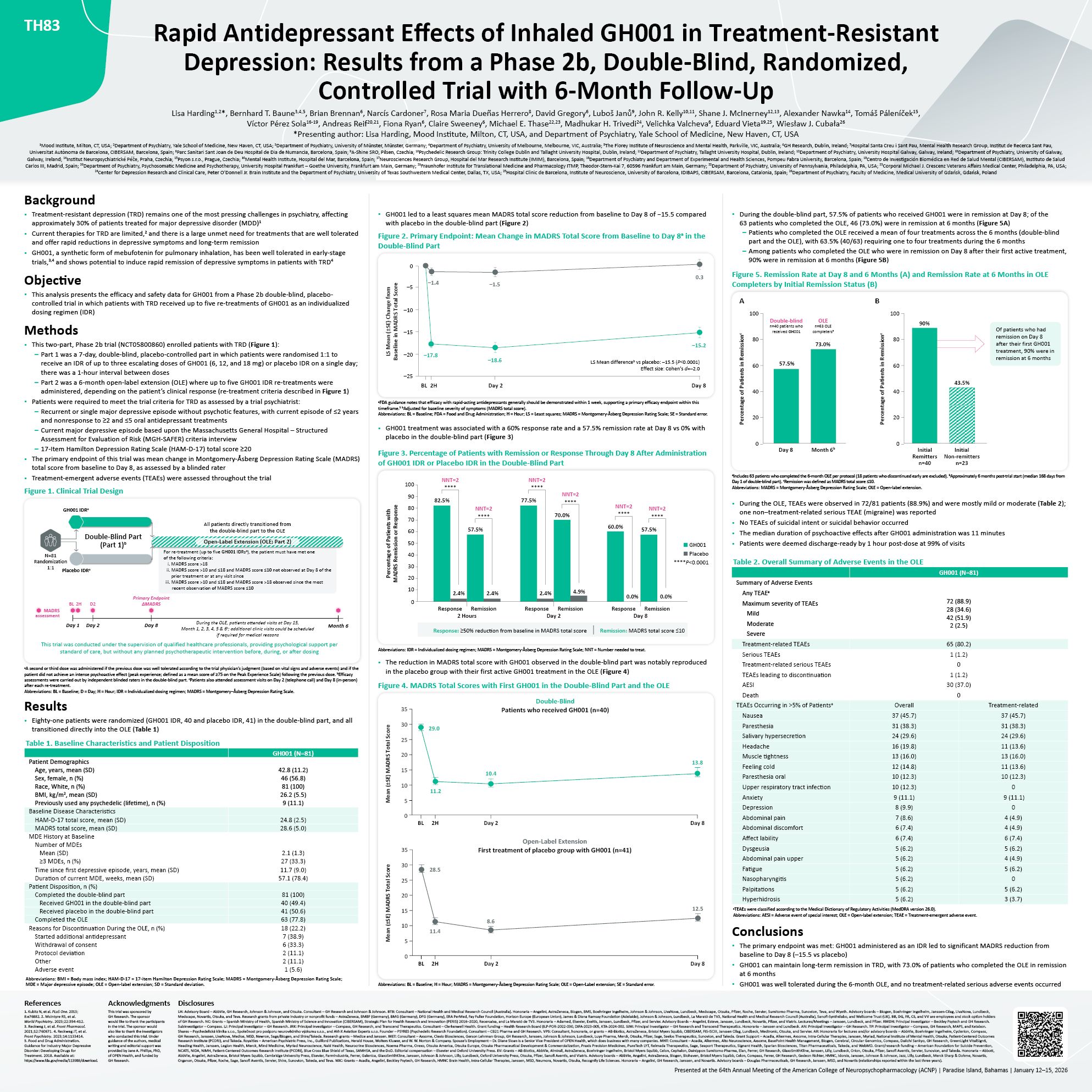
GH Research PLC announces the presentation of posters related to its GH001-TRD-201 and GH001-BD-202 clinical trials at the 64th American College of Neuropsychopharmacology (ACNP) annual
meeting (the “Congress”), which is scheduled to take place from January 12-15, 2026, in Paradise Island, Bahamas.
A copy of the poster to be presented by Sanjay J. Mathew during the Congress is attached hereto as Exhibit 99.1.
A copy of the poster to be presented by Lisa Harding during the Congress is attached hereto as Exhibit 99.2.
A copy of the poster to be presented by Andreas Reif during the Congress is attached hereto as Exhibit 99.3.
1
EXHIBIT INDEX
|
Poster to be presented by Sanjay J. Mathew with Title: Suicidal Ideation and Behavior in Patients with Treatment-Resistant Depression Treated with GH001
|
|
|
Poster to be presented by Lisa Harding with Title: Rapid Antidepressant Effects of Inhaled GH001 in Treatment-Resistant Depression: Results from a Phase 2b,
Double-Blind, Randomized Controlled Trial with 6-Month Follow-Up
|
|
|
Poster to be presented by Andreas Reif with Title: Results of a Phase 2a Clinical Trial of Inhaled Mebufotenin (GH001) in Patients with Bipolar II Disorder and a
Current Major Depressive Episode
|
2
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
GH Research PLC
|
||
|
Date: January 8, 2025
|
||
|
By:
|
/s/ Julie Ryan
|
|
|
Name:
|
Julie Ryan
|
|
|
Title:
|
Vice President, Finance
|
|
3
Exhibit 99.1

62.5 77.5 70.7 68.3 22.5 10.0 12.2 12.2 12.5 5.0 5.0 14.6 17.1 Figure 3. Double-Blind Part: MADRS Suicidality Item 10 Responses at Baseline and Day 8 GH001 (n=40) Placebo (n=41) 6 5 4 3 2 1 0 100 90 80 70 60 50 40 30 20 10 0 Baseline Day 8 Baseline Day 8 Percentage of Patients by MADRS Suicidality Item Score 2.5 2.5 2.4 2.4 Higher scores indicate worse symptoms. Abbreviation: MADRS = Montgomery–Åsberg Depression Rating Scale. In the 6-month OLE, the numbers of patients reporting suicidal ideation according to the C-SSRS at all timepoints assessed during the trial were lower than before the first GH001 treatment period; C-SSRS items endorsed during each active treatment period are shown in Figure 4 Figure 4. Proportion of Patients Reporting Suicidal Ideationa by C-SSRS Item in Each GH001 Treatment Period in the Double-Blind Part and the OLE 8.2 10.2 16.0 6.2 2.5 10.0 4.3 1.4 7.7 7.7 100 90 80 70 60 50 40 30 20 10 0 Predose Day 8 Active TP1 Predose Day 8 Active TP2 5.0 5.0 Predose Day 8 Active TP3 Predose Day 8 Active TP4 Predose Day 8 Active TP5 Percentage of Patients Indicating Yes on C-SSRS Items 1–3 1.7 1.7 3.8 Suicidal ideation Q3 Suicidal ideation Q2 Suicidal ideation Q1 aPatients responding “yes” to C-SSRS items 1 (wish to be dead), 2 (non-specific active suicidal thoughts), or 3 (active suicidal ideation with any methods); no patients responded “yes” post-dose to items 4 (active suicidal ideation with some intent) or 5 (active suicidal ideation with specific plan). Abbreviations: C-SSRS = Columbia-Suicide Severity Rating Scale; Q = Question; TP = Treatment period. During the OLE, the proportions of patients reporting a score of zero on MADRS item 10 at all timepoints during GH001 treatment were greater than that at baseline of the double-blind part (Figure 5) – The median (range) change from baseline to Month 6 in MADRS score for item 10 was 0.0 (–3 to 2; n=74) Figure 5. MADRS Suicidality Item 10 Responses at Baseline and by Month in the OLE 6 5 4 3 2 1 0 66.7 73.2 88.3 83.6 89.3 89.5 76.1 17.3 11.3 6.7 13.1 5.4 8.8 12.7 13.6 12.7 7.0 1.2 1.2 1.4 1.4 5.0 1.6 1.6 5.4 1.4 2.8 1.8 100 90 80 70 60 50 40 30 20 10 0 Month 5 Month 6/ET Baseline Day 28/Month 1 Month 2 Month 3 Month 4 Percentage of Patients by MADRS Suicidality Item Score Higher scores indicate worse symptoms. Abbreviations: ET = End of treatment; MADRS = Montgomery–Åsberg Depression Rating Scale; OLE = Open-label extension. No TEAEs of suicidal intent or suicidal behavior occurred throughout the 6-month duration of the trial A TEAE of suicidal ideation occurred in one patient; the event lasted 6 hours before resolving spontaneously – This TEAE was not accompanied by any changes in C-SSRS score beyond the duration for which the thoughts occurred, and the patient did not report any further TEAEs of suicidal ideation during the trial Conclusions In this double-blind, placebo-controlled trial with a 6-month OLE in patients with TRD who were not at acute risk of suicide at baseline, GH001 was not associated with treatment-emergent events of suicidal ideation with intent or suicidal behavior, or with any increased suicidal ideation as measured by the C-SSRS immediately following treatment or over time for up to 6 months These results support that GH001 was generally well tolerated and was associated with significant reductions in depressive symptoms without increasing risk of suicide • Treatment-resistant depression (TRD) is a debilitating condition that has been defined as a failure to respond to ≥2 adequate treatments for major depressive disorder (MDD)1 Rates of suicidal ideation in patients with TRD are higher than in patients with treatment-responsive MDD (15% vs 6% of patients, respectively)2 – TRD is also associated with higher rates of attempted suicide, all-cause mortality, and deaths by suicide3 There is a significant unmet need for safe and effective therapies for TRD that do not exacerbate suicidal intent In a Phase 2b trial, GH001, a synthetic form of mebufotenin (5-MeO-DMT) for pulmonary inhalation, was well tolerated and resulted in rapid and significant improvements in depressive symptoms in patients with TRD Objective We describe the safety effects of GH001 on suicidal ideation and behavior in patients with TRD enrolled in the Phase 2b trial Methods • This trial (NCT05800860) included a 7-day, randomized, double-blind part and a 6-month open-label extension (OLE; Figure 1) In the double-blind part, patients were randomized 1:1 to receive an individualized dosing regimen (IDR) of up to three escalating doses of GH001 (6, 12, and 18 mg) or placebo IDR on a single day with a 1-hour interval between doses In the OLE, patients received up to five GH001 IDR re-treatments over 6 months as needed, depending on the patient’s clinical response and tolerability of previous IDRs Trial eligibility criteria excluded patients with suicidal ideation with intent (with items 4 or 5 on the Columbia- Suicide Severity Rating Scale [C-SSRS] endorsed) within the past year, during the screening period, or at baseline; those with suicidal behaviors or non-suicidal self-injury in the past year; and those with a clinical assessment of significant suicide risk Suicidal behavior and suicidal ideation were assessed using the C-SSRS at screening; during each treatment period at pre-dose, discharge, Day 2, and Day 8; and at all OLE scheduled visits (Day 15 and monthly up to Month 6/end of treatment) The C-SSRS “baseline/screening” version was used at screening, and the “since last visit” version was used at all subsequent visits Suicidal ideation was defined as “yes” responses to items 1–5 (with items 4 and 5 indicating suicidal ideation with intent), and suicidal behavior was defined as “yes” responses to items 6–10 Montgomery–Åsberg Depression Rating Scale (MADRS) item 10 (suicidal thoughts) was used to provide further quantification of suicidal ideation, which was indicated by scores ≥2 on a scale from 0–64 Treatment-emergent adverse events (TEAEs) were assessed at each visit Results were analyzed descriptively Figure 1. Clinical Trial Schematic For re-treatment (up to five GH001 IDRsa), the patient must have met one of the following criteria: MADRS score >18 MADRS score >10 and ≤18 and MADRS score ≤10 not observed at Day 8 of the prior treatment or at any visit since MADRS score >10 and ≤18 and MADRS score >18 observed since the most recent observation of MADRS score ≤10 N=81 Randomization 1:1 GH001 IDRa Placebo IDRa Primary Endpoint ΔMADRS Double-Blind Part (Part 1)b Day 1 Day 8 During the OLE, patients attended visits at Day 15, Month 1, 2, 3, 4, 5 & 6c; additional clinic visits could be scheduled if required for medical reasons MADRS assessment Month 6 BL 2H D2 Day 2 This trial was conducted under the supervision of qualified healthcare professionals, providing psychological support per standard of care, but without any planned psychotherapeutic intervention before, during, or after dosing All patients directly transitioned from the double-blind part to the OLE Open-Label Extension (OLE; Part 2) aA second or third dose was administered if the previous dose was well tolerated according to the trial physician’s judgment (based on vital signs and adverse events) and if the patient did not achieve an intense psychoactive effect (peak experience; defined as a mean score of ≥75 on the Peak Experience Scale) following the previous dose. bEfficacy assessments were carried out by independent blinded raters in the double-blind part. cPatients also attended assessment visits on Day 2 (telephone call) and Day 8 (in-person) after each re-treatment. Abbreviations: BL = Baseline; D = Day; H = Hour; IDR = Individualized dosing regimen; MADRS = Montgomery–Åsberg Depression Rating Scale. TH71 Background Results Suicidal Ideation and Behavior in Patients with Treatment-Resistant Depression Treated with GH001 Sanjay J. Mathew1,*, Bernhard T. Baune2-4, Narcís Cardoner5, Wiesław J. Cubała6, Kelly Doolin7, Rosa Maria Dueñas Herrero8, David Gregory7, Luboš Janů9, John R. Kelly10,11, Rachael MacIssac7, Shane J. McInerney12,13, Alexander Nawka14, Tomáš Páleníček15, Víctor Pérez Sola16-19, Andreas Reif20,21, Claire Sweeney7, Madhukar H. Trivedi22, Velichka Valcheva7, Eduard Vieta19,23, Michael E. Thase24,25 *Presenting author: Sanjay J. Mathew, Department of Psychiatry and Behavioral Sciences, Texas A&M University Naresh K. Vashisht College of Medicine, Bryan, TX, USA 1Department of Psychiatry and Behavioral Sciences, Texas A&M University Naresh K. Vashisht College of Medicine, Bryan, TX, USA; 2Department of Psychiatry, University of Münster, Münster, Germany; 3Department of Psychiatry, University of Melbourne, Melbourne, VIC, Australia; 4The Florey Institute of Neuroscience and Mental Health, Parkville, VIC, Australia; 5Hospital Santa Creu i Sant Pau, Mental Health Research Group, Institut de Recerca Sant Pau, Universitat Autònoma de Barcelona, CIBERSAM, Barcelona, Spain; 6Department of Psychiatry, Faculty of Medicine, Medical University of Gdańsk, Gdańsk, Poland; 7GH Research, Dublin, Ireland; 8Parc Sanitari Sant Joan de Deu Hospital de Dia de Numancia, Barcelona, Spain; 9A-Shine SRO, Pilsen, Czechia; 10Psychedelic Research Group: Trinity College Dublin and Tallaght University Hospital, Dublin, Ireland; 11Department of Psychiatry, Tallaght University Hospital, Dublin, Ireland; 12Department of Psychiatry, University Hospital Galway, Galway, Ireland; 13Department of Psychiatry, University of Galway, Galway, Ireland; 14Institut Neuropsychiatrické Péče, Praha, Czechia; 15Psyon s.r.o., Prague, Czechia; 16Mental Health Institute, Hospital del Mar, Barcelona, Spain; 17Neurosciences Research Group, Hospital del Mar Research Institute (IMIM), Barcelona, Spain; 18Department of Psychiatry and Department of Experimental and Health Sciences, Pompeu Fabra University, Barcelona, Spain; 19Centro de Investigación Biomédica en Red de Salud Mental (CIBERSAM), Instituto de Salud Carlos III, Madrid, Spain; 20Department of Psychiatry, Psychosomatic Medicine and Psychotherapy, University Hospital Frankfurt – Goethe University, Frankfurt am Main, Germany; 21Fraunhofer Institute for Translational Medicine and Pharmacology ITMP, Theodor-Stern-Kai 7, 60596, Frankfurt am Main, Germany; 22Center for Depression Research and Clinical Care, Peter O’Donnell Jr. Brain Institute and the Department of Psychiatry, University of Texas Southwestern Medical Center, Dallas, TX, USA; 23Hospital Clinic de Barcelona, Institute of Neuroscience, University of Barcelona, IDIBAPS, CIBERSAM, Barcelona, Catalonia, Spain; 24Department of Psychiatry, University of Pennsylvania, Philadelphia, PA, USA; 25Corporal Michael J. Crescenz Veterans Affairs Medical Center, Philadelphia, PA, USA References McIntyre RS, et al. World Psychiatry. 2023;22:394-412. Mrazek DA, et al. Psychiatr Serv. 2014;65:977-87. Kern DM, et al. Brain Behav. 2023;13:e3171. Montgomery SA, Asberg M. Br J Psychiatry. 1979;134:382-9. Acknowledgments This trial was sponsored by GH Research. The sponsor would like to thank the participants in the trial. The sponsor would also like to thank the investigators who conducted this trial. Under guidance of the authors, medical writing and editorial support was provided by Jane A. Phillips, PhD, of OPEN Health, and funded by GH Research. Disclosures SJMa: Consultant − Autobahn, Boehringer-Ingelheim, Clexio Biosciences, Engrail Therapeutics, EMA Wellness, Engrail Therapeutics, Motif Neurotech, Neumora, Newleos, Relmada Therapeutics, Signant Health, Supernus, and Xenon Pharmaceuticals. Research support − Abbott, Boehringer Ingelheim, and Engrail Therapeutics. BTB: Consultant – National Health and Medical Research Council (Australia). Honoraria – Angelini, AstraZeneca, Biogen, BMS, Boehringer Ingelheim, Johnson & Johnson, LivaNova, Lundbeck, Medscape, Otsuka, Pfizer, Roche, Servier, Sumitomo Pharma, Sunovion, Teva, and Wyeth. Advisory boards – Biogen, Boehringer Ingelheim, Janssen-Cilag, LivaNova, Lundbeck, Medscape, Novartis, Otsuka, and Teva. Research grants from private industries or nonprofit funds – AstraZeneca, BMBF (Germany), BMG (Germany), DFG (Germany), ERA PerMed, Fay Fuller Foundation, Horizon Europe (European Union), James & Diana Ramsay Foundation (Adelaide), Johnson & Johnson, Lundbeck, La Marató de TV3, National Health and Medical Research Council (Australia), Sanofi-Synthélabo, and Wellcome Trust (UK). NC: Grants – Spanish Ministry of Health, Spanish Ministry of Science and Innovation (CIBERSAM), Strategic Plan for Health Research and Innovation (PERIS) 2016–2020, Recercaixa, and La Marató de TV3. Honoraria – Adamed, Elsevier, Exeltis, Janssen, Lundbeck, Pfizer, and Servier. Advisory boards – Angelini, Esteve, Janssen, Lundbeck, Novartis, Pfizer, and Viatris. Lectures/meetings – Janssen, Lundbeck, and Pfizer. WJC: Grants – Acadia, Angelini, Beckley Psytech, GH Research, HMNC Brain Health, Intra-Cellular Therapies, Janssen, MSD, Neumora, Novartis, Otsuka, and Recognify Life Sciences. Honoraria – Angelini, GH Research, Janssen, and Novartis. Advisory boards – Douglas Pharmaceuticals, GH Research, Janssen, MSD, and Novartis. KD, DG, RM, CS, and VV are employees and shareholders of GH Research. RMDH: Principal investigator – Beckley Psytech and GH Research. Subinvestigator – Compass. LJ: Principal investigator – GH Research. JRK: Principal investigator – Compass, GH Research, and Transcend Therapeutics. Consultant – Clerkenwell Health. Grant funding – Health Research Board (ILP-POR-2022-030, DIFA-2023-005, KTA-2024-002). SJMc: Principal investigator – GH Research and Transcend Therapeutics. Honoraria – Janssen and Lundbeck. AN: Principal investigator – GH Research. TP: Principal investigator – Compass, GH Research, MAPS, and Ketabon. Shares – Psychedelická klinika s.r.o., Společnost pro podporu neurovědního výzkumu s.r.o., and AVI-X Aviation Experts s.r.o. Founder – PSYRES (Psychedelic Research Foundation). Consultant – CB21 Pharma and GH Research. VPS: Consultant, honoraria, or grants – AB-Biotics, AstraZeneca, Bristol Myers Squibb, CIBERSAM, FIS-ISCiii, Janssen Cilag, Lundbeck, Medtronic, Otsuka, and Servier. AR: Honoraria for lectures and/or advisory boards – AbbVie, Boehringer Ingelheim, Cyclerion, Compass, GH Research, Janssen, LivaNova, Medice, MSD, Newron, Sage/Biogen, and Shire/Takeda. Research grants – Medice and Janssen. MHT: Consultant – Acadia, Alkermes, Alto Neuroscience, Axsome, BasePoint Health Management, Biogen, Cerebral, Circular Genomics, Compass, Daiichi Sankyo, GH Research, GreenLight VitalSign6, Heading Health, Janssen, Legion Health, Merck, Mind Medicine, Myriad Neuroscience, Naki Health, Neurocrine Biosciences, Noema Pharma, Orexo, Otsuka America, Otsuka Europe, Otsuka Pharmaceutical Development & Commercialization, Praxis Precision Medicines, PureTech LYT, Relmada Therapeutics, Sage, Seaport Therapeutics, Signant Health, Sparian Biosciences, Titan Pharmaceuticals, Takeda, and WebMD. Grant/research funding – American Foundation for Suicide Prevention, NCATS, NIDA, NIMH, Patient-Centered Outcomes Research Institute (PCORI), Blue Cross Blue Shield of Texas, SAMHSA, and the DoD. Editorial compensation – Elsevier and Oxford University Press. EV: Grants – AB-Biotics, AbbVie, Almirall, AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Celon, Cephalon, Dainippon Sumitomo Pharma, Elan, Ferrer, GH Research, GlaxoSmithKline, Janssen, Lilly, Lundbeck, Orion, Otsuka, Pfizer, Sanofi Aventis, Servier, Sunovion, and Takeda. Honoraria – Abbott, AbbVie, Angelini, AstraZeneca, Bristol Myers Squibb, Cambridge University Press, Elsevier, Farmindustria, Ferrer, Galenica, GlaxoSmithKline, Janssen, Johnson & Johnson, Lilly, Lundbeck, Oxford University Press, Otsuka, Pfizer, Sanofi Aventis, and Viatris. Advisory boards – AbbVie, Angelini, AstraZeneca, Biogen, Biohaven, Bristol Myers Squibb, Celon, Compass, Ferrer, GH Research, Gedeon Richter, HMNC, Idorsia, Janssen, Johnson & Johnson, Jazz, Lilly, Lundbeck, Merck Sharp & Dohme, Novartis, Organon, Otsuka, Pfizer, Roche, Sage, Sanofi Aventis, Servier, Shire, Sunovion, Takeda, and Teva. MET: Consultant – Axsome, Clexio Biosciences, Gerson Lehrman Group, GH Research, Janssen, Johnson & Johnson, Lundbeck, Luye Pharma, Merck, Otsuka, Pfizer, Sage, Seelos Therapeutics, Sunovion, and Takeda. Grant support – Acadia, Alkermes, Axsome, Intra-Cellular Therapies, Janssen, Myriad, National Institute of Mental Health, Otsuka, Patient-Centered Outcomes Research Institute (PCORI), and Takeda. Royalties – American Psychiatric Press, Inc., Guilford Publications, Herald House, Wolters Kluwer, and W. W. Norton & Company. Spouse’s employment – Dr. Diane Sloan is a Senior Vice President of OPEN Health, which does business with many companies. Presented at the 64th Annual Meeting of the American College of Neuropsychopharmacology (ACNP) | Paradise Island, Bahamas | January 12–15, 2026 GH001 (n=40) Placebo (n=41) Eighty-one patients were randomized (GH001 IDR, 40 and placebo IDR, 41) in the double-blind part, and all transitioned directly into the OLE Rates of historic and current suicidal ideation were balanced across treatment groups (Table 1) Table 1. Baseline Demographics and Clinical Characteristics Patient Demographics Age, years, mean (SD) 41.6 (11.4) 43.9 (10.9) Sex, female, n (%) 24 (60.0) 22 (53.7) Race, White, n (%) 40 (100) 41 (100) Actively employed, n (%) 33 (82.5) 29 (70.7) Disease Characteristics HAM-D-17 total score, mean (SD) 24.9 (2.6) 24.6 (2.3) MADRS total score, mean (SD) 29.0 (5.4) 28.2 (4.6) CGI-S, mean (SD) 4.8 (0.7) 5.0 (0.6) Suicidality Non-suicidal self-injurious behavior Lifetime, n (%) 1 (2.5) 0 Past 12 months, n (%) 0 0 Baseline, n (%) 0 0 Suicidal ideation without intenta Lifetime, n (%) 15 (37.5) 23 (56.1) Past 12 months, n (%) 10 (25.0) 18 (43.9) Baseline, n (%) 7 (17.5) 9 (22.0) Suicidal ideation with intenta Past 12 months, n (%) 0 0 Baseline, n (%) 0 0 Suicidal behaviora Past 12 months, n (%) 0 0 Baseline, n (%) 0 0 aSuicidal ideation without intent was defined as any “yes” answer to C-SSRS items 1–3; suicidal ideation with intent was defined as any “yes” answer to C-SSRS items 4–5; suicidal behavior was defined as any “yes” answer to C-SSRS items 6–10; patients were counted for each category item to which they answered “yes” and therefore may be counted in more than one item. Abbreviations: CGI-S = Clinical Global Impression–Severity; C-SSRS = Columbia-Suicide Severity Rating Scale; HAM-D-17 = Hamilton Rating Scale for Depression; MADRS = Montgomery–Åsberg Depression Rating Scale; SD = Standard deviation. • In the GH001 group, suicidal ideation was reported by seven patients at baseline and four at Day 8 (Figure 2) No GH001-treated patients developed new-onset suicidal ideation between baseline and Day 8 In the placebo group, five patients reported suicidal ideation both at baseline and at Day 8, two reported suicidal ideation at baseline but not Day 8, and two patients without baseline suicidal ideation reported it at Day 8 No suicidal behaviors were reported in GH001- or placebo-treated patients at baseline or on any scheduled C-SSRS assessments in the double-blind part 82.5 17.5 90.0 7.5 82.9 82.9 2.5 2.4 2.4 14.6 14.6 Percentage of Patients Indicating Yes Versus No on C-SSRS Items 1–3 Baseline Day 8 Baseline Day 8 Figure 2. Double-Blind Part: Proportion of Patients with and without Suicidal Ideationa at Baseline and Day 8 by C-SSRS Item GH001 (n=40) Placebo (n=41) 0 20 40 50 60 70 90 30 10 80 100 Suicidal ideation Q3 Suicidal ideation Q2 Suicidal ideation Q1 No suicidal ideation aPatients responding “yes” to C-SSRS items 1 (wish to be dead), 2 (non-specific active suicidal thoughts), or 3 (active suicidal ideation with any methods); no patients responded “yes” post-dose to items 4 (active suicidal ideation with some intent) or 5 (active suicidal ideation with specific plan). Abbreviations: C-SSRS = Columbia-Suicide Severity Rating Scale; Q = Question. Baseline MADRS item 10 (suicidal thoughts) scores were similar between the two treatment groups, and patients who received GH001 did not report worsening of symptoms on Day 8 of the double-blind part as per MADRS item 10 (Figure 3) – The median (range) change from baseline to Day 8 in MADRS score for item 10 was 0.0 (–2 to 1) for patients who received GH001 and 0.0 (–1 to 2) for patients who received placebo
Exhibit 99.2
Presented at the 64th Annual Meeting of the American College of
Neuropsychopharmacology (ACNP) | Paradise Island, Bahamas | January 12–15, 2026 1Mood Institute, Milton, CT, USA; 2Department of Psychiatry, Yale School of Medicine, New Haven, CT, USA; 3Department of Psychiatry, University of Münster,
Münster, Germany; 4Department of Psychiatry, University of Melbourne, Melbourne, VIC, Australia; 5The Florey Institute of Neuroscience and Mental Health, Parkville, VIC, Australia; 6GH Research, Dublin, Ireland; 7Hospital Santa Creu i
Sant Pau, Mental Health Research Group, Institut de Recerca Sant Pau, Universitat Autònoma de Barcelona, CIBERSAM, Barcelona, Spain; 8Parc Sanitari Sant Joan de Deu Hospital de Dia de Numancia, Barcelona, Spain; 9A-Shine SRO, Pilsen,
Czechia; 10Psychedelic Research Group: Trinity College Dublin and Tallaght University Hospital, Dublin, Ireland; 11Department of Psychiatry, Tallaght University Hospital, Dublin, Ireland; 12Department of Psychiatry, University Hospital
Galway, Galway, Ireland; 13Department of Psychiatry, University of Galway, Galway, Ireland; 14Institut Neuropsychiatrické Péče, Praha, Czechia; 15Psyon s.r.o., Prague, Czechia; 16Mental Health Institute, Hospital del Mar, Barcelona,
Spain; 17Neurosciences Research Group, Hospital del Mar Research Institute (IMIM), Barcelona, Spain; 18Department of Psychiatry and Department of Experimental and Health Sciences, Pompeu Fabra University, Barcelona, Spain; 19Centro de
Investigación Biomédica en Red de Salud Mental (CIBERSAM), Instituto de Salud Carlos III, Madrid, Spain; 20Department of Psychiatry, Psychosomatic Medicine and Psychotherapy, University Hospital Frankfurt – Goethe University, Frankfurt am
Main, Germany; 21Fraunhofer Institute for Translational Medicine and Pharmacology ITMP, Theodor-Stern-Kai 7, 60596 Frankfurt am Main, Germany; 22Department of Psychiatry, University of Pennsylvania, Philadelphia, PA, USA; 23Corporal
Michael J. Crescenz Veterans Affairs Medical Center, Philadelphia, PA, USA; 24Center for Depression Research and Clinical Care, Peter O’Donnell Jr. Brain Institute and the Department of Psychiatry, University of Texas Southwestern
Medical Center, Dallas, TX, USA; 25Hospital Clinic de Barcelona, Institute of Neuroscience, University of Barcelona, IDIBAPS, CIBERSAM, Barcelona, Catalonia, Spain; 26Department of Psychiatry, Faculty of Medicine, Medical University of
Gdańsk, Gdańsk, Poland Rapid Antidepressant Effects of Inhaled GH001 in Treatment-Resistant Depression: Results from a Phase 2b, Double-Blind, Randomized, Controlled Trial with 6-Month Follow-Up Lisa Harding1,2*, Bernhard T. Baune3,4,5,
Brian Brennan6, Narcís Cardoner7, Rosa Maria Dueñas Herrero8, David Gregory6, Luboš Janů9, John R. Kelly10,11, Shane J. McInerney12,13, Alexander Nawka14, Tomáš Páleníček15, Víctor Pérez Sola16-19, Andreas Reif20,21, Fiona Ryan6, Claire
Sweeney6, Michael E. Thase22,23, Madhukar H. Trivedi24, Velichka Valcheva6, Eduard Vieta19,25, Wiesław J. Cubała26 *Presenting author: Lisa Harding, Mood Institute, Milton, CT, USA, and Department of Psychiatry, Yale School of Medicine,
New Haven, CT, USA References 1. Kubitz N, et al. PLoS One. 2013; 8:e76882. 2. McIntyre RS, et al. World Psychiatry. 2023;22:394-412. 3. Reckweg J, et al. Front Pharmacol. 2021;12:760671. 4. Reckweg JT, et al. Front Psychiatry.
2023;14:1133414. 5. Food and Drug Administration. Guidance for Industry Major Depressive Disorder: Developing Drugs for Treatment. 2018. Available at: https://www.fda.gov/media/113988/download. Acknowledgments This trial was sponsored
by GH Research. The sponsor would like to thank the participants in the trial. The sponsor would also like to thank the investigators who conducted this trial. Under guidance of the authors, medical writing and editorial support was
provided by Jane A. Phillips, PhD, of OPEN Health, and funded by GH Research. Disclosures LH: Advisory Board – AbbVie, GH Research, Johnson & Johnson, and Otsuka. Consultant – GH Research and Johnson & Johnson. BTB: Consultant –
National Health and Medical Research Council (Australia). Honoraria – Angelini, AstraZeneca, Biogen, BMS, Boehringer Ingelheim, Johnson & Johnson, LivaNova, Lundbeck, Medscape, Otsuka, Pfizer, Roche, Servier, Sumitomo Pharma,
Sunovion, Teva, and Wyeth. Advisory boards – Biogen, Boehringer Ingelheim, Janssen-Cilag, LivaNova, Lundbeck, Medscape, Novartis, Otsuka, and Teva. Research grants from private industry or nonprofit funds – AstraZeneca, BMBF (Germany),
BMG (Germany), DFG (Germany), ERA PerMed, Fay Fuller Foundation, Horizon Europe (European Union), James & Diana Ramsay Foundation (Adelaide), Johnson & Johnson, Lundbeck, La Marató de TV3, National Health and Medical Research
Council (Australia), Sanofi-Synthélabo, and Wellcome Trust (UK). BB, DG, FR, CS, and VV are employees and stock option holders of GH Research. NC: Grants – Spanish Ministry of Health, Spanish Ministry of Science and Innovation (CIBERSAM),
Strategic Plan for Health Research and Innovation (PERIS) 2016–2020, Recercaixa, and La Marató de TV3. Honoraria – Adamed, Elsevier, Exeltis, Janssen, Lundbeck, Pfizer, and Servier. Advisory Boards – Angelini, Esteve, Janssen, Lundbeck,
Novartis, Pfizer, and Viatris. Lectures/Meetings – Janssen, Lundbeck, and Pfizer. RMDH: Principal investigator – Beckley Psytech and GH Research. Subinvestigator – Compass. LJ: Principal investigator – GH Research. JRK: Principal
investigator – Compass, GH Research, and Transcend Therapeutics. Consultant – Clerkenwell Health. Grant funding – Health Research Board (ILP-POR-2022-030, DIFA-2023-005, KTA-2024-002. SJM: Principal investigator – GH Research and
Transcend Therapeutics. Honoraria – Janssen and Lundbeck. AN: Principal investigator – GH Research. TP: Principal investigator – Compass, GH Research, MAPS, and Ketabon. Shares – Psychedelická klinika s.r.o., Společnost pro podporu
neurovědního výzkumu s.r.o., and AVI-X Aviation Experts s.r.o. Founder – PSYRES (Psychedelic Research Foundation). Consultant – CB21 Pharma and GH Research. VPS: Consultant, honoraria, or grants – AB-Biotics, AstraZeneca, Bristol Myers
Squibb, CIBERSAM, FIS-ISCIII, Janssen Cilag, Lundbeck, Medtronic, Otsuka, and Servier. AR: Honoraria for lectures and/or advisory boards – AbbVie, Boehringer Ingelheim, Cyclerion, Compass, GH Research, Janssen, LivaNova, Medice, MSD,
Newron, Sage/Biogen, and Shire/Takeda. Research grants – Medice and Janssen. MET: Consultant – Axsome, Clexio Biosciences, Gerson Lehrman Group, GH Research, Janssen, Johnson & Johnson, Lundbeck, Luye Pharma, Merck, Otsuka, Pfizer,
Sage, Seelos Therapeutics, Sunovion, and Takeda. Grant Support – Acadia, Alkermes, Axsome, Intra-Cellular Therapies, Janssen, Myriad, National Institute of Mental Health, Otsuka, Patient-Centered Outcomes Research Institute (PCORI), and
Takeda. Royalties – American Psychiatric Press, Inc., Guilford Publications, Herald House, Wolters Kluwer, and W. W. Norton & Company. Spouse’s Employment – Dr. Diane Sloan is a Senior Vice President of OPEN Health, which does
business with many companies. MHT: Consultant – Acadia, Alkermes, Alto Neuroscience, Axsome, BasePoint Health Management, Biogen, Cerebral, Circular Genomics, Compass, Daiichi Sankyo, GH Research, GreenLight VitalSign6, Heading Health,
Janssen, Legion Health, Merck, Mind Medicine, Myriad Neuroscience, Naki Health, Neurocrine Biosciences, Noema Pharma, Orexo, Otsuka America, Otsuka Europe, Otsuka Pharmaceutical Development & Commercialization, Praxis Precision
Medicines, PureTech LYT, Relmada Therapeutics, Sage, Seaport Therapeutics, Signant Health, Sparian Biosciences, Titan Pharmaceuticals, Takeda, and WebMD. Grant/research funding – American Foundation for Suicide Prevention, NCATS, NIDA,
NIMH, Patient-Centered Outcomes Research Institute (PCORI), Blue Cross Blue Shield of Texas, SAMHSA, and the DoD. Editorial compensation – Elsevier and Oxford University Press. EV: Grants – AB-Biotics, AbbVie, Almirall, AstraZeneca,
Boehringer Ingelheim, Bristol Myers Squibb, Celon, Cephalon, Dainippon Sumitomo Pharma, Elan, Ferrer, GH Research, GlaxoSmithKline, Janssen, Lilly, Lundbeck, Orion, Otsuka, Pfizer, Sanofi Aventis, Servier, Sunovion, and Takeda. Honoraria
– Abbott, AbbVie, Angelini, AstraZeneca, Bristol Myers Squibb, Cambridge University Press, Elsevier, Farmindustria, Ferrer, Galenica, GlaxoSmithKline, Janssen, Johnson & Johnson, Lilly, Lundbeck, Oxford University Press, Otsuka,
Pfizer, Sanofi Aventis, and Viatris. Advisory boards – AbbVie, Angelini, AstraZeneca, Biogen, Biohaven, Bristol Myers Squibb, Celon, Compass, Ferrer, GH Research, Gedeon Richter, HMNC, Idorsia, Janssen, Johnson & Johnson, Jazz, Lilly,
Lundbeck, Merck Sharp & Dohme, Novartis, Organon, Otsuka, Pfizer, Roche, Sage, Sanofi Aventis, Servier, Shire, Sunovion, Takeda, and Teva. WJC: Grants – Acadia, Angelini, Beckley Psytech, GH Research, HMNC Brain Health, Intra-Cellular
Therapies, Janssen, MSD, Neumora, Novartis, Otsuka, Recognify Life Sciences. Honoraria – Angelini, GH Research, Janssen, and Novartis. Advisory boards – Douglas Pharmaceuticals, GH Research, Janssen, MSD, and Novartis (relationships
reported within the last three years). Background • • Treatment-resistant depression (TRD) remains one of the most pressing challenges in psychiatry, affecting approximately 30% of patients treated for major depressive disorder
(MDD)1 Current therapies for TRD are limited,2 and there is a large unmet need for treatments that are well tolerated and offer rapid reductions in depressive symptoms and long-term remission GH001, a synthetic form of mebufotenin for
pulmonary inhalation, has been well tolerated in early-stage trials,3,4 and shows potential to induce rapid remission of depressive symptoms in patients with TRD4 Objective This analysis presents the efficacy and safety data for GH001
from a Phase 2b double-blind, placebo- controlled trial in which patients with TRD received up to five re-treatments of GH001 as an individualized dosing regimen (IDR) Methods • • • This two-part, Phase 2b trial (NCT05800860)
enrolled patients with TRD (Figure 1): Part 1 was a 7-day, double-blind, placebo-controlled part in which patients were randomised 1:1 to receive an IDR of up to three escalating doses of GH001 (6, 12, and 18 mg) or placebo IDR on a
single day; there was a 1-hour interval between doses Part 2 was a 6-month open-label extension (OLE) where up to five GH001 IDR re-treatments were administered, depending on the patient’s clinical response (re-treatment criteria
described in Figure 1) Patients were required to meet the trial criteria for TRD as assessed by a trial psychiatrist: Recurrent or single major depressive episode without psychotic features, with current episode of ≤2 years and
nonresponse to ≥2 and ≤5 oral antidepressant treatments Current major depressive episode based upon the Massachusetts General Hospital – Structured Assessment for Evaluation of Risk (MGH-SAFER) criteria interview 17-Item Hamilton
Depression Rating Scale (HAM-D-17) total score ≥20 The primary endpoint of this trial was mean change in Montgomery-Åsberg Depression Rating Scale (MADRS) total score from baseline to Day 8, as assessed by a blinded
rater Treatment-emergent adverse events (TEAEs) were assessed throughout the trial Figure 1. Clinical Trial Design N=81 Randomization 1:1 GH001 IDRa Placebo IDRa Double-Blind Part (Part 1)b Day 8 During the OLE, patients
attended visits at Day 15, Month 1, 2, 3, 4, 5 & 6c; additional clinic visits could be scheduled if required for medical reasons MADRS assessment Month 6 Primary Endpoint BL 2H D2 ΔMADRS Day 1 Day 2 This trial was conducted
under the supervision of qualified healthcare professionals, providing psychological support per standard of care, but without any planned psychotherapeutic intervention before, during, or after dosing All patients directly transitioned
from the double-blind part to the OLE Open-Label Extension (OLE; Part 2) For re-treatment (up to five GH001 IDRsa), the patient must have met one of the following criteria: MADRS score >18 MADRS score >10 and ≤18 and MADRS score
≤10 not observed at Day 8 of the prior treatment or at any visit since MADRS score >10 and ≤18 and MADRS score >18 observed since the most recent observation of MADRS score ≤10 aA second or third dose was administered if the
previous dose was well tolerated according to the trial physician’s judgment (based on vital signs and adverse events) and if the patient did not achieve an intense psychoactive effect (peak experience; defined as a mean score of ≥75 on
the Peak Experience Scale) following the previous dose. bEfficacy assessments were carried out by independent blinded raters in the double-blind part. cPatients also attended assessment visits on Day 2 (telephone call) and Day 8
(in-person) after each re-treatment. Abbreviations: BL = Baseline; D = Day; H = Hour; IDR = Individualized dosing regimen; MADRS = Montgomery–Åsberg Depression Rating Scale. Results Eighty-one patients were randomized (GH001 IDR, 40
and placebo IDR, 41) in the double-blind part, and all transitioned directly into the OLE (Table 1) Table 1. Baseline Characteristics and Patient Disposition GH001 (N=81) Patient Demographics Age, years, mean (SD) 42.8 (11.2) Sex,
female, n (%) 46 (56.8) Race, White, n (%) 81 (100) BMI, kg/m2, mean (SD) 26.2 (5.5) Previously used any psychedelic (lifetime), n (%) 9 (11.1) Baseline Disease Characteristics HAM-D-17 total score, mean (SD) 24.8 (2.5) MADRS
total score, mean (SD) 28.6 (5.0) MDE History at Baseline Number of MDEs Mean (SD) 2.1 (1.3) ≥3 MDEs, n (%) 27 (33.3) Time since first depressive episode, years, mean (SD) 11.7 (9.0) Duration of current MDE, weeks, mean (SD) 57.1
(78.4) Patient Disposition, n (%) Completed the double-blind part 81 (100) Received GH001 in the double-blind part Received placebo in the double-blind part 40 (49.4) 41 (50.6) Completed the OLE 63 (77.8) Reasons for
Discontinuation During the OLE, n (%) 18 (22.2) Started additional antidepressant 7 (38.9) Withdrawal of consent 6 (33.3) Protocol deviation 2 (11.1) Other 2 (11.1) Adverse event 1 (5.6) GH001 led to a least squares mean MADRS
total score reduction from baseline to Day 8 of −15.5 compared with placebo in the double-blind part (Figure 2) Figure 2. Primary Endpoint: Mean Change in MADRS Total Score from Baseline to Day 8a in the Double-Blind
Part 0 –5 –10 –15 –25 LS Mean (±SE) Change from Baseline in MADRS Total Score LS Mean differenceb vs placebo: –15.5 (P<0.0001) Effect size: Cohen’s d=–2.0 0.3 –15.2 –1.5 –18.6 –1.4 –20 –17.8 BL 2H Day 2 Day 8 aFDA
guidance notes that efficacy with rapid-acting antidepressants generally should be demonstrated within 1 week, supporting a primary efficacy endpoint within this timeframe.5 bAdjusted for baseline severity of symptoms (MADRS total
score). Abbreviations: BL = Baseline; FDA = Food and Drug Administration; H = Hour; LS = Least squares; MADRS = Montgomery-Åsberg Depression Rating Scale; SE = Standard error. GH001 treatment was associated with a 60% response rate and
a 57.5% remission rate at Day 8 vs 0% with placebo in the double-blind part (Figure 3) Figure 3. Percentage of Patients with Remission or Response Through Day 8 After Administration of GH001 IDR or Placebo IDR in the Double-Blind
Part Percentage of Patients with MADRS Remission or Response 82.5% NNT=2 **** 100 90 80 70 60 50 40 30 20 10 0 2.4%
2.4% 57.5% NNT=2 **** 2.4% 77.5% NNT=2 **** 4.9% 70.0% **** NNT=2 60.0% NNT=2 **** Response Remission Response Remission Response Remission 2 Hours Day 2 Day 8 0.0%
0.0% 57.5% NNT=2 **** GH001 Placebo ****P<0.0001 Response: ≥50% reduction from baseline in MADRS total score | Remission: MADRS total score ≤10 Abbreviations: IDR = Individualized dosing regimen; MADRS = Montgomery-Åsberg
Depression Rating Scale; NNT = Number needed to treat. The reduction in MADRS total score with GH001 observed in the double-blind part was notably reproduced in the placebo group with their first active GH001 treatment in the OLE (Figure
4) Figure 4. MADRS Total Scores with First GH001 in the Double-Blind Part and the OLE BL 2H Day 2 Day 8 35 30 25 20 15 29.0 10 11.2 5 0 10.4 13.8 28.5 11.4 8.6 12.5 BL 2H Day 2 Day
8 35 30 25 20 15 10 5 0 Mean (±SE) MADRS Total Score Mean (±SE) MADRS Total Score Double-Blind Patients who received GH001 (n=40) Open-Label Extension First treatment of placebo group with GH001 (n=41) Abbreviations: BMI =
Body mass index; HAM-D-17 = 17-item Hamilton Depression Rating Scale; MADRS = Montgomery-Åsberg Depression Rating Scale; MDE = Major depressive episode; OLE = Open-label extension; SD = Standard deviation. Abbreviations: BL = Baseline; H
= Hour; MADRS = Montgomery-Åsberg Depression Rating Scale; OLE = Open-Label extension; SE = Standard error. • During the double-blind part, 57.5% of patients who received GH001 were in remission at Day 8; of the 63 patients who
completed the OLE, 46 (73.0%) were in remission at 6 months (Figure 5A) Patients who completed the OLE received a mean of four treatments across the 6 months (double-blind part and the OLE), with 63.5% (40/63) requiring one to four
treatments during the 6 months Among patients who completed the OLE who were in remission on Day 8 after their first active treatment, 90% were in remission at 6 months (Figure 5B) Percentage of Patients in
Remissionc 57.5% 90% 43.5% Double-blind n=40 patients who received GH001 OLE n=63 OLE completersa Day 8 Month 6b Initial Initial Remitters Non-remitters n=40 n=23 Figure 5. Remission Rate at Day 8 and 6 Months (A) and Remission
Rate at 6 Months in OLE Completers by Initial Remission Status (B) A B Percentage of Patients in Remissionc 60 40 20 0 100 100 80 80 73.0% 60 40 20 0 Of patients who had remission on Day 8 after their first GH001 treatment,
90% were in remission at 6 months aIncludes 63 patients who completed the 6-month OLE per protocol (18 patients who discontinued early are excluded). bApproximately 6 months post-trial start (median 168 days from Day 1 of double-blind
part). cRemission was defined as MADRS total score ≤10. Abbreviations: MADRS = Montgomery-Åsberg Depression Rating Scale; OLE = Open-label extension. During the OLE, TEAEs were observed in 72/81 patients (88.9%) and were mostly mild or
moderate (Table 2); one non–treatment-related serious TEAE (migraine) was reported No TEAEs of suicidal intent or suicidal behavior occurred The median duration of psychoactive effects after GH001 administration was 11 minutes Patients
were deemed discharge-ready by 1 hour post-dose at 99% of visits TEAEs Occurring in >5% of Patientsa Overall Treatment-related Nausea 37 (45.7) 37 (45.7) Paresthesia 31 (38.3) 31 (38.3) Salivary hypersecretion 24 (29.6) 24
(29.6) Headache 16 (19.8) 11 (13.6) Muscle tightness 13 (16.0) 13 (16.0) Feeling cold 12 (14.8) 11 (13.6) Paresthesia oral 10 (12.3) 10 (12.3) Upper respiratory tract infection 10 (12.3) 0 Anxiety 9 (11.1) 9
(11.1) Depression 8 (9.9) 0 Abdominal pain 7 (8.6) 4 (4.9) Abdominal discomfort 6 (7.4) 4 (4.9) Affect lability 6 (7.4) 6 (7.4) Dysgeusia 5 (6.2) 5 (6.2) Abdominal pain upper 5 (6.2) 4 (4.9) Fatigue 5 (6.2) 5
(6.2) Nasopharyngitis 5 (6.2) 0 Palpitations 5 (6.2) 5 (6.2) Hyperhidrosis 5 (6.2) 3 (3.7) Table 2. Overall Summary of Adverse Events in the OLE GH001 (N=81) Summary of Adverse Events Any TEAEa Maximum severity of TEAEs
Mild Moderate Severe 72 (88.9) 28 (34.6) 42 (51.9) 2 (2.5) Treatment-related TEAEs 65 (80.2) Serious TEAEs Treatment-related serious TEAEs TEAEs leading to discontinuation AESI Death 1 (1.2) 0 1 (1.2) 30 (37.0) 0 aTEAEs
were classified according to the Medical Dictionary of Regulatory Activities (MedDRA version 26.0). Abbreviations: AESI = Adverse event of special interest; OLE = Open-label extension; TEAE = Treatment-emergent adverse
event. Conclusions • The primary endpoint was met: GH001 administered as an IDR led to significant MADRS reduction from baseline to Day 8 (–15.5 vs placebo) GH001 can maintain long-term remission in TRD, with 73.0% of patients who
completed the OLE in remission at 6 months GH001 was well tolerated during the 6-month OLE, and no treatment-related serious adverse events occurred • • TH83
Exhibit 99.3

Dose 1 6 mg GH001 Dose 2a 12 mg GH001 Dose 3a 18 mg GH001 1-hour
interval Single Day IDR 1-hour interval N = 6 Patients with BDII and a current MDE Day-1 End-of-trial MADRS Safety Screening Pre-test day Day 8 Day 1 Day-60 to -2 Day 2 GH001 Follow-up Administration MADRS MADRS Safety
Safety N=6 Completed trial, n (%) 6 (100) Discontinued, n (%) 0 Number of previous MDE, mean (SD) 14.0 (12.4) Duration of current MDE (weeks), mean (SD) 20.8 (22.7) MADRS total score at baseline, mean (SD) 32.0
(5.1) Demographics Female, n (%) 4 (66.7) Age (years), mean (SD) 44.2 (9.3) Height (cm), mean (SD) 174.7 (10.1) Weight (kg), mean (SD) 76.1 (18.6) BMI (kg/m2), mean (SD) 24.8 (5.0) Race, White, n (%) 6 (100) Background Bipolar
II disorder (BDII) is a chronic psychiatric disorder characterized by alternating episodes of hypomania and major depressive episodes (MDE), imposing high burdens of illness on individuals1 The estimated lifetime prevalence rate of BDII is
between 0.4 and 5%2,3 Current treatments for depressive symptoms in patients with BDII remain limited, offering insufficient efficacy and tolerability highlighting the need for new therapeutic approaches4 Mebufotenin (5-MeO-DMT) is a rapid
acting psychoactive molecule that acts as a non-selective serotonin agonist with highest affinity for the 5-HT receptor subtype5 1A GH001, a synthetic form of mebufotenin for pulmonary inhalation, has been well tolerated in early-stage
trials and has shown potential to induce rapid remission of depressive symptoms in patients with treatment-resistant depression (TRD)6,7 The trial presented here is the first in which mebufotenin was administered to patients diagnosed with
BDII and a current MDE Objective To investigate the safety and antidepressant effects of GH001 in adult patients with BDII and a current MDE Methods This Phase 2a, proof-of-concept, open-label trial (NCT05839509) enrolled patients aged
18-64 years who met Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) diagnostic criteria for BDII with a current MDE Patients were required to have a Montgomery–Åsberg Depression Rating Scale (MADRS) total score of
≥24 and a Young Mania Rating Scale (YMRS) total score of ≤8 at baseline and prior to dosing on Day 1 Patients were not permitted to receive any antidepressant medications within 7 days or 5 half-lives, whichever was longer, prior to dosing.
Lithium use within 6 months prior to dosing was not permitted, if applicable Patients were administered an individualized dosing regimen (IDR) of up to three escalating doses of GH001 (6, 12, and 18 mg) with a 1-hour interval between doses
on a single day (Figure 1) This trial was conducted under the supervision of qualified healthcare professionals, providing psychological support per standard-of-care, but without any planned psychotherapeutic intervention before, during, or
after dosing The primary endpoint was change in MADRS total score from baseline to Day 8 Secondary endpoints included response (≥50% reduction from baseline in MADRS total score), remission (MADRS total score ≤10), Clinical Global
Impression-Severity (CGI-S) scale, Bipolar Depression Rating Scale (BDRS), and safety and tolerability Figure 1. Clinical Trial Design References American Psychiatric Association, DSM-5 Task Force. (2013). Merikangas KR, et al. Arch Gen
Psychiatry, 2011;68(3):241-251. Benazzi F. CNS Drugs, 2007;21(9):727-40. Aaronson ST, et al. JAMA Psych. 2024;81(6):555-562. Halberstadt AL, et al. Psychopharmacology. 2012;221:709-718. Reckweg J, et al. Front Pharmacol.
2021;12:760671. Reckweg JT, et al. Front Psychiatry. 2023;14:1133414. Acknowledgments This trial was sponsored by GH Research. The sponsor would like to thank the participants in the trial. The sponsor would also like to thank the
investigators who conducted the trial. Under guidance of authors, medical writing and editorial support was provided by Brian Brennan, PhD of GH Research Ireland Limited. Statistical analysis was carried out by Rachael MacIsaac, PhD, of GH
Research. Disclosures AR: Honoraria for lectures and/or advisory boards – AbbVie, Boehringer Ingelheim, Cyclerion, Compass, GH Research, Janssen, LivaNova, Medice, MSD, Newron, Sage/Biogen, and Shire/Takeda. Research grants – Medice and
Janssen. MB: Advisor to Alfred E. Tiefenbacher GmbH Co. KG, COMPASS Pathfinder Ltd., GH Research, MedEd-Link Inc., Janssen Global Services, LLC, Livanova, Mindforce Game Lab AB, and Novartis. Received lecture fees from MedTrix GmbH and
Streamedup GmbH. MDL and PR: Nothing to disclose. FD: Consultant to GH Research. KK, POG, VV: Employee and stock option holder of GH Research. CBS: Shareholder of GH Research. FD: Consultant to GH Research. MET: Grants – Acadia, Alkermes,
Axsome, Intra-Cellular Therapies, Janssen, National Institute of Mental Health, Otsuka, Patient-Centered Outcomes Research Institute (PCORI), and Takeda. Advisory Boards – Autobahn Therapeutics, Axsome, Clexio Biosciences, Gerson Lehrman
Group, GH Research, Lundbeck, Janssen, Johnson & Johnson, Luye Pharma, Merck, Object Pharma, Otsuka, Pfizer, Sage, Seelos Therapeutics, Sunovion, and Takeda. Royalties – American Psychiatric Association Foundation, Guilford Publications,
Herald House, Wolters Kluwer, and W W Norton & Company. BTB: Consultant – National Health and Medical Research Council (Australia). Honoraria – Angelini, AstraZeneca, Biogen, BMS, Boehringer Ingelheim, Johnson & Johnson, LivaNova,
Lundbeck, Medscape, Otsuka, Pfizer, Roche, Servier, Sumitomo Pharma, Sunovion, Teva, Viatris, and Wyeth. Advisory boards – Biogen, Boehringer Ingelheim, Janssen-Cilag, LivaNova, Lundbeck, Medscape, Novartis, GH Research, Otsuka, and Teva.
Research grants from private industries or nonprofit funds – AstraZeneca, BMBF (Germany), BMG (Germany), DFG (Germany), ERA PerMed, Fay Fuller Foundation, Horizon Europe (European Union), James & Diana Ramsay Foundation (Adelaide),
Johnson & Johnson, Lundbeck, La Marató de TV3, National Health and Medical Research Council (Australia), Sanofi-Synthélabo, and Wellcome Trust (UK). Presented at the 64th Annual Meeting of the American College of Neuropsychopharmacology
(ACNP) Nassau, Bahamas | January 12–15, 2026 TU82 Conclusions In this trial evaluating the safety and antidepressant effects of GH001 in patients with BDII and a current MDE, the primary endpoint was met: a significant reduction from
baseline in MADRS total score was observed on Day 8 Significant reductions in MADRS total scores were also observed at 2-hours post-dose, supporting the rapid onset of antidepressant effects of GH001 GH001 administered via inhalation
demonstrated a favorable safety profile and was well tolerated in patients with BDII and a current MDE; no treatment-related SAEs were reported aA second or third dose was administered if the previous dose was well tolerated according to the
trial physician’s judgement (based on vital signs and adverse events) and if the patient did not achieve an intense psychoactive effect (peak experience; defined as a mean score of ≥75 on the Peak Experience Scale) following the previous
dose. Abbreviations: BDII = Bipolar II disorder; IDR = Individualized dosing regimen; MADRS = Montgomery–Åsberg Depression Rating Scale; MDE = Major depressive episode. Results Disposition and Demographics A total of six patients with BDII
and a current MDE were enrolled in this trial. Patient disposition and demographics are presented in Table 1 Table 1. Patient Disposition and Baseline Characteristics Results of a Phase 2a Clinical Trial of Inhaled Mebufotenin (GH001) in
Patients with Bipolar II Disorder and a Current Major Depressive Episode Andreas Reif1,2, Michael Bauer3, Max de Leeuw4, Fabian Devlin5, Katerina Kriger6, Padraig O’Grady6, Philipp Ritter3,7, Claus Bo Svendsen6, Michael E. Thase8,9, Velichka
Valcheva6, Bernhard T. Baune10,11,12 1Department of Psychiatry, Psychosomatic Medicine and Psychotherapy, University Hospital Frankfurt – Goethe University, Frankfurt am Main, Germany; 2Fraunhofer Institute for Translational Medicine and
Pharmacology ITMP, Theodor-Stern-Kai 7, 60596, Frankfurt am Main, Germany; 3Department of Psychiatry and Psychotherapy, Faculty of Medicine, University Hospital Carl Gustav Carus, Dresden, Germany; 4Department of Psychiatry, Leiden University
Medical Center, Leiden, The Netherlands; 5Mental Health Research for Innovation Centre, Mersey Care NHS Foundation Trust, United Kingdom; 6GH Research, Dublin, Ireland; 7Institute of Psychiatry, Psychology and Neuroscience, King’s College
London, United Kingdom; 8Department of Psychiatry, University of Pennsylvania, Philadelphia, PA, USA; 9Corporal Michael J. Crescenz Veterans Affairs Medical Center, Philadelphia, PA, USA; 10Department of Psychiatry, University of Münster,
Münster, Germany; 11Department of Psychiatry, University of Melbourne, Melbourne, VIC, Australia; 12The Florey Institute of Neuroscience and Mental Health, Parkville, VIC, Australia. Abbreviations: BMI = Body mass index; MADRS =
Montgomery–Åsberg Depression Rating Scale; MDE = Major depressive episode; SD = Standard deviation. Efficacy The primary endpoint was achieved, with a significant reduction from baseline to Day 8 in mean (standard deviation [SD]) MADRS
total score of −16.8 (12.2) (P=0.0099; Figure 2) Significant reductions in mean (SD) MADRS total score were also observed at 2-hours post-dose (–16.3 [6.0]) and Day 2 (–13.3 [13.5]; Figure 2) One-third (33.3%) of patients responded and were
in remission on Day 8 The rapid reduction in the severity of depressive symptoms as assessed by the MADRS, were mirrored in the CGI-S and the BDRS A mean (SD) reduction of –2.5 (1.5) on the CGI-S and –14.5 (11.2) on the BDRS were observed
from baseline to Day 8 (Figure 3) Figure 2. Mean Change in MADRS Total Score From Baseline in Patients With BDII and a Current MDE Figure 3. Mean Change in BDRS Total Score From Baseline in Patients With BDII and a Current
MDE -18 -16 -14 -12 -10 -8 -6 -4 -2 0 Mean (SD) Change in MADRS from Baseline Day 2 -13.3 (13.5) P<0.0299 Baseline Day 8 -16.8 (12.2) P=0.0099 2-hours post-dose -16.3 (6.0) P<0.0006 Mean (SD) Change in BDRS from
Baseline Day 2 -13.5 (11.8) Day 8 -14.5 (11.2) 0 -2 -4 -6 -8 -10 -12 -14 -16 Baseline Abbreviations: BDII = Bipolar II disorder; MADRS = Montgomery–Åsberg Depression Rating Scale; MDE = Major depressive episode; SD = Standard
deviation. Safety Abbreviations: BDII = Bipolar II disorder; BL = Baseline; BDRS = Bipolar Depression Rating Scale; MDE = Major depressive episode; SD = Standard deviation. Treatment-emergent adverse events (TEAEs) were observed in 5/6
patients (83.3%) and were mostly mild in severity (87.5%) with two moderate and one severe events Headache (50%), nausea (33.3%) and anxiety (33.3%) were the most frequently reported TEAEs; all other TEAEs occurred in a single patient
each One severe event of anxiety was reported which subsided within 24 hours; no TEAEs of flashbacks were reported There were no treatment-emergent serious adverse events (SAEs), and no patient withdrew from the trial There were no
clinically significant changes in spirometry after inhalation of GH001 There was a clinically significant reduction in mean (SD) Brief Psychiatric Rating Scale from baseline to Day 8 (−15.7 [12.0]). There was no clinically relevant worsening
of other clinician-rated assessments (based on the Clinical Assessment of Discharge Readiness, Columbia-Suicide Severity Rating Scale, and Modified Observer’s Assessment of Alertness and Sedation scales) and all patients were deemed ready for
discharge within the same day of dosing Following dosing with GH001, YMRS scores remained low and stable, decreasing from 2.2 at baseline to 1.0 by Day 8 (−1.2 [SD=1.5]), indicating no emergence of manic symptoms The safety profile observed
in this trial was consistent with other completed trials investigating GH001 in TRD and postpartum depression Table 2. Summary of Safety in Patients with BDII and a Current MDE Event # n (%) Any TEAE 18 5 (83.3) Mild 15 5
(83.3) Moderate 2 2 (33.3) Severe 1 1 (16.7) Treatment-related TEAEs 18 5 (83.3) Treatment-emergent SAE 0 0 TEAEs by Preferred Term Headache 4 3 (50.0) Nausea 6 2 (33.3) Anxiety 2 2 (33.3) Paresthesia 1 1
(16.7) Agitation 1 1 (16.7) Hypoaesthesia oral 1 1 (16.7) Neck pain 1 1 (16.7) Fatigue 1 1 (16.7) Cough 1 1 (16.7) Abbreviations: BDII = Bipolar type II; MDE = Major depressive episode; SAE = Serious adverse event; TEAE =
Treatment-emergent adverse event.
